
To participate in the reaction with others sulphur initially loses these four electrons and will get +4 oxidation state. There are four electrons in the 3p orbital and 2 electrons in 3s orbital in the sulphur. The electronic configuration of sulphur is as follows. The atomic number of sulphur is 16 means there are 16 electrons present in sulphur. Sulphur is an element which is present in the VI A group in the periodic table and belongs to p-block. Oxidation state also termed as oxidation number. 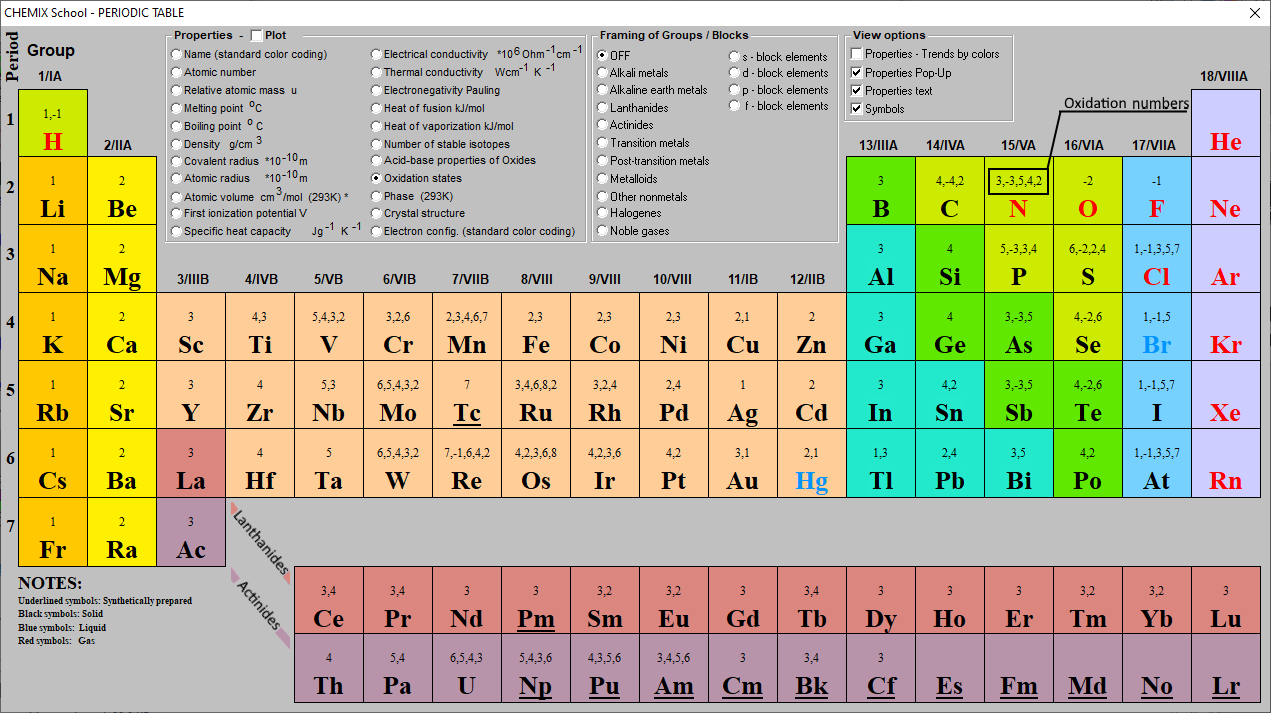
In the question it is asked what the common oxidation state of sulphur is. Oxidation number will be positive or negative depending on the losing or gaining of electrons. But the oxidation number of an element is going to vary from one compound to another and depends on the chemical reaction in which it participates. Since the oxidation state of copper has reduced from +2 to 0, this is a reduction reaction.Hint: Oxidation number or oxidation state is the charge present on the atom or on the molecule. The two electrons released from the zinc atom are accepted by the copper ion, in the corresponding reduction reaction: Since zinc has changed its oxidation state from 0 to +2, this is an oxidation reaction. In this reaction, the mini-reactions of oxidation and reduction are as follows: For instance, let’s consider the standard metal displacement reaction, Now that you know this, you will be able to do much better at defining the confusing terms of oxidation and reduction in terms of the oxidation number. These were the important rules about determining oxidation numbers. Turning the compound into an algebraic equation in this fashion is the best way of figuring out the oxidation number of unknown elements. For instance, if we consider the charged ion SO 4 2-, which has a charge of -2, the equation turns out to be: Whereas the previous rule dealt with electrically stable compounds, this rule deals with charged compounds, but on the same principle. Rule #8: Just like the previous rule, the net oxidation number of a polyatomic ion is equal to the charge on it. Incidentally, it is also found in the +4 and +2 oxidation states. Thus, for H 2SO 4 to be balanced, sulfur’s oxidation number has to be +6, which is one of its oxidation states. For instance, if we consider the stable compound H 2SO 4 (sulfuric acid), we know the net oxidation number of hydrogen and oxygen in this compound to be: This is helpful in figuring out the oxidation number of a particular element in a compound. 
Rule #7: The sum of the oxidation numbers of all components of a neutral compound is zero. Like the previous rule, this is not valid if the metals are in elemental form, in which case the first rule supersedes. Rule #6: Alkaline earth metals such as magnesium and potassium have an oxidation number of +2. The only time this changes is when the metals are in the elemental form, in which case their oxidation state is zero. Rule #5: Lying in the same group of the periodic table as hydrogen, alkali metals such as lithium, sodium, and potassium also have an oxidation number of +1. The question of oxidation states only applies to reactions where ionic bonds are formed. The bonds between atoms of the same element are always covalent, wherein electrons are shared, rather than ionic, wherein electrons are passed from one reagent to another. This means that O 2 (oxygen), Mg 2 (magnesium), Al (aluminum), He (helium), and S 8 (sulfur) have the oxidation number zero, because the oxidation state of the individual atoms wasn’t changed during the formation of the molecule. 
This is true regardless of whether the element exists as an atom or a polyatomic molecule. Rule #1: The oxidation state of an uncombined element not possessing an electric charge is always zero. Now that we have a rough idea of what an oxidation number is, let’s move on to the rules of determining the oxidation number of a particular substance. For instance, in the aforementioned reaction between hydrogen and chlorine, the loss of electrons adds to the oxidation state of the hydrogen atoms, which become positively charged ions, while the gain of electrons reduces the oxidation state of the chlorine atoms, which become negatively charged ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
